By Kelly Grayson
Waveform capnography is an example of EMS as an early adopter of emerging technology. Often, in the hospital, recognition of the value of capnography has not spread beyond the confines of the anesthesiology department. As a patient advocate, use your knowledge of capnography to communicate important patient information to emergency department and intensive care unit staff.
You’re picking up a patient in the ED, for a critical care transfer to the ICU at a Level I trauma center an hour away. Your patient has a subarachnoid hemorrhage, and was intubated in the ED and placed on a propofol infusion for sedation. Vent settings are assist/control mode, FiO2 of 40 percent, tidal volume of 650 mL, PEEP of 5.0 cm H20, and ventilatory rate of 16.
Your patient’s hemodynamics are within acceptable parameters, and he seems to be tolerating the ventilator well, but when you attach your transport ventilator and end-tidal capnography adaptor, you see what you had suspected; your patient’s ETCO2 is only 20 mm Hg, well below physiologic norms. A quick perusal of the patient’s chart reveals arterial blood gases drawn five minutes after the patient was intubated an hour ago, and none taken since.
So when you hand off your patient to the accepting ICU staff at the trauma center, how do you justify your decision to adjust the vent settings to a more appropriate rate of 12 breaths/minute?
If you’ve ever dropped off an intubated, post-ROSC patient with an ETCO2 of 40 mm Hg at a ventilatory rate of 10 breaths/minute, only to see the ED staff immediately double your ventilatory rate under the erroneous assumption that all post-arrest patients are acidotic, or had an ED nurse look at you like you’ve sprouted a second head when you say you didn’t administer a bronchodilator to a known asthma patient because both your capnograph and your stethoscope confirmed that there was no evidence of bronchospasm, then you know that not every ED staffer understands capnography like we do.
But if you’ll follow these capnography documentation and handoff tips, you’ll paint a much clearer picture of the patient’s condition, and do a better job of explaining how capnography findings relate to that condition.
Which patients need capnography?
There is an old medical truism that states, “If you never check a temperature, you’re going to miss a lot of fevers.” The same philosophy holds true for quantitative waveform capnography.
When we understand that capnography is the only real-time measure of the effectiveness of ventilation and a reliable indirect measure of perfusion, then that opens up an entire new world of applications. Simply put, we should be using capnography on any patient with significantly decreased level of consciousness, or any significant derangement in oxygenation, ventilation, or perfusion.
That adds up to a lot of patients.
Intubated and mechanically ventilated patients
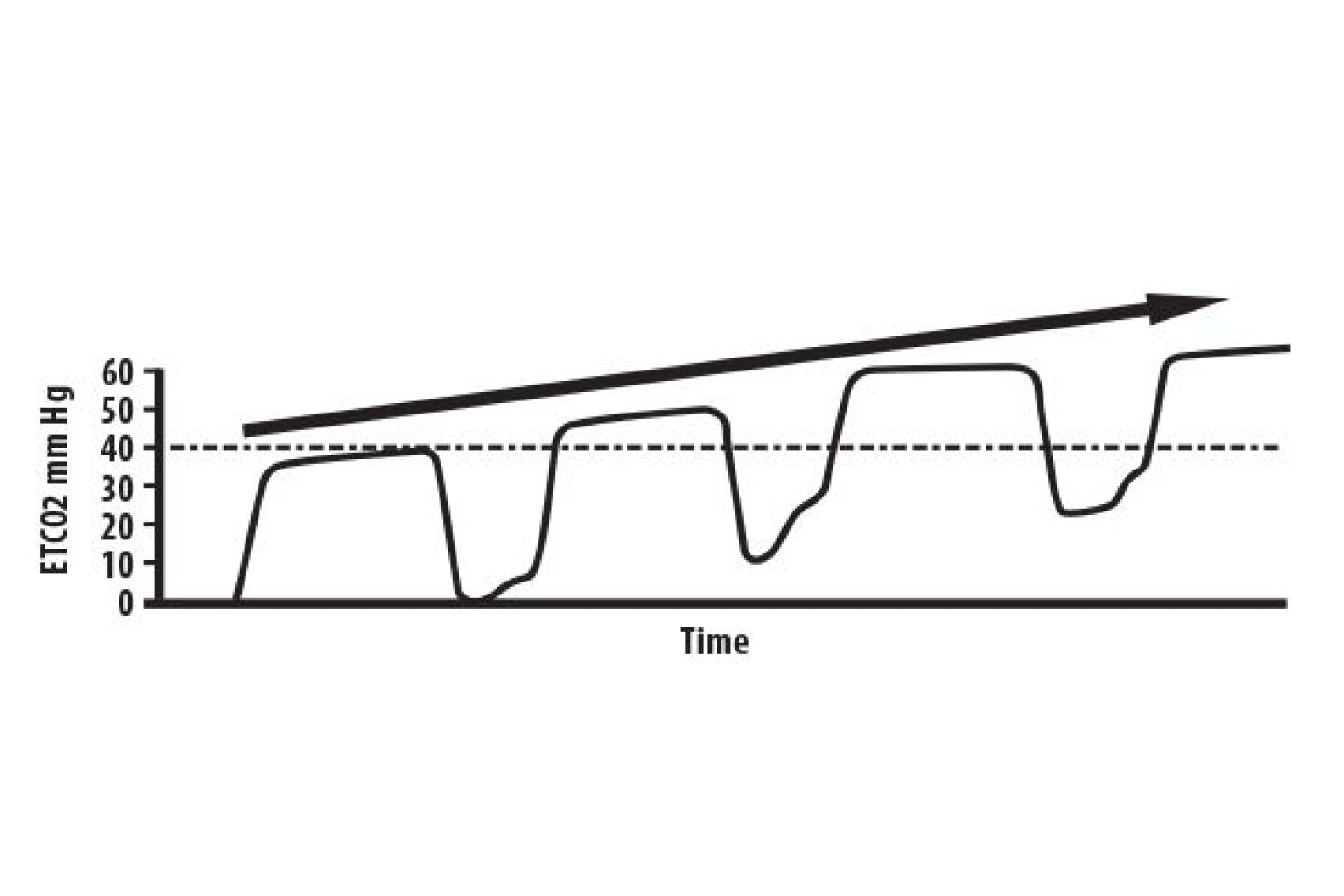
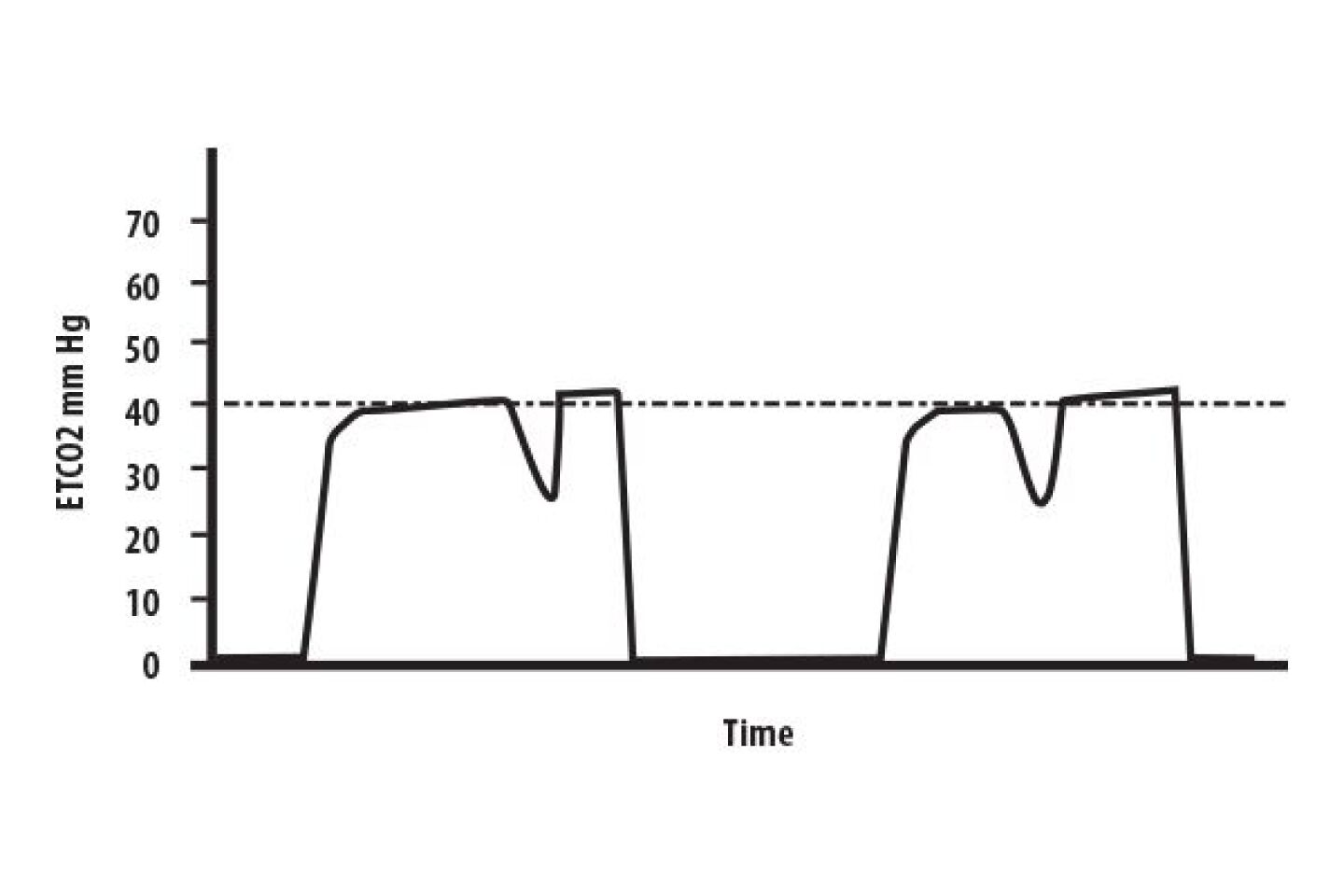
Just like with normal breathing, ventilatory rate in mechanically ventilated patients has a direct bearing on ETCO2. When attaching a capnograph to an intubated, mechanically ventilated patient, there are four parameters to consider: initial ETCO2 reading, waveform morphology, ventilatory rate, and respiratory baseline (Phase I) measurement.
In patients with normal pulmonary physiology, end-tidal carbon dioxide (PetCO2) should be no more than 2-5 mm Hg less than arterial carbon dioxide (PaCO2), and is usually within 1 mm Hg of PaCO2 [1]. Document initial PetCO2 readings, and compare those to arterial blood gases, if any are available. Normal PaCO2 is 35-45 mm Hg, therefore PetCO2 would theoretically be 34-44 mm Hg, and should never be more than 5 mm Hg less than the arterial CO2 in patients with normal pulmonary physiology.
However, most intubated patients do not have normal pulmonary physiology, or they wouldn’t have been intubated in the first place. Use caution in changing ventilator rate settings to titrate ventilations to a normal PetCO2 reading. If the patient has significant ventilation/perfusion (V/Q) mismatch due to pulmonary shunting, you risk hypoventilating the patient when titrating ventilations to PetCO2.
If the end-tidal gradient, P(a-et)CO2 is more than 10 mm Hg, a shunt exists, but it is impossible to determine this unless you are comparing capnography readings and arterial blood gases measured within the same time frame. When documenting a change in ventilator rate settings and why, be sure to include PaCO2 and PetCO2 and the time each was measured (ideally, within the same time frame), and the change in PetCO2 with the new settings.
Pay particular attention to the inspiratory baseline (Phase 1) in mechanically ventilated patients. Ideally, the baseline should be zero. If the baseline is above zero in a mechanically ventilated patient and the Beta angle is greater than 90 degrees, this is an indication that the patient is rebreathing carbon dioxide.
Incompetent inspiratory or expiratory valves or a depleted CO2 absorber may be the culprit. If your ventilator isn’t flashing an alarm and a troubleshooting message, briefly ventilate the patient with a BVM on room air, and recheck the inspiratory baseline. If it returns to zero, you have a malfunctioning ventilator.
Sudden change in the capnography waveform (prolonged Phase III, increased Beta angle) and a decreased PetCO2 may indicate pulmonary embolus, pneumothorax, or migration of the tube into the right mainstem, resulting in single lung ventilation.
In patients with chest tubes, inability to maintain an alveolar plateau, with “stair step” Phase 0, may indicate occlusion of the chest tube [2,3].
Patients who have undergone neuromuscular blockade may exhibit inspiratory effort at a level too low to trigger the ventilator as the neuromuscular blockers wear off. This “curare cleft,” named after the first primitive neuromuscular blocker, presents as a miniscule notch in the alveolar plateau (Phase III).
When you notice curare clefts in your capnography waveform, you only have 2-3 minutes to re-administer the neuromuscular blockade before the patient comes out of paralysis.
Intra and post-arrest patients
During cardiopulmonary resuscitation, waveform capnography can serve a variety of valuable functions. As a prognostic indicator of resuscitation outcomes, PetCO2 that is consistently lower than 10 mm Hg despite adequate ventilation and effective chest compressions can be considered a criterion for termination of resuscitation efforts [4, 5]. If protocols dictate consultation with medical control before terminating resuscitation in the field, be sure to inform the physician that CO2 levels have been consistently less than 10 mm Hg, and include the PetCO2 reading at termination of resuscitation efforts in your documentation.
During resuscitation, effectiveness of chest compressions can be reflected in the capnography waveforms [6]. Diminishing PetCO2 can indicate compressor fatigue, or if there is a significant disparity in PetCO2 readings between rescuers, a flaw in one rescuer’s compression technique.
A sudden increase in PetCO2 during resuscitation may indicate return of spontaneous circulation (ROSC), and often precedes a palpable peripheral pulse. This has been demonstrated in animal models as well as human cardiac arrest patients [7,8,9].
Always recheck and document PetCO2 after any major move in packaging and transport, and the PetCO2 at time of handoff in the Emergency Department. Titrate ventilation rate to a physiologically normal PetCO2 (35-45 mm Hg), and communicate the PetCO2 and rate required to achieve it to the ED staff at handoff.
Respiratory patients
Waveform capnography can be a useful monitoring parameter in asthma patients, and may help differentiate the wheezes of cardiac asthma from the obstructive bronchospasm characteristic of asthma and other obstructive pulmonary diseases. While wheezes may be present in early stages of acute pulmonary edema, the patients usually lack the characteristic “shark fin” waveform of acute bronchospasm.
While monitoring non-intubated respiratory patients, carefully monitor the inspiratory baseline, waveform morphology and PetCO2. Rising carbon dioxide readings may indicate respiratory fatigue. Elevated inspiratory baseline may indicate rebreathing of CO2, either from too low an oxygen flow rate, or from malfunction of the valve in a non-rebreather mask.
PetCO2 may be decreased in tachypneic patients with pulmonary embolus, but may be differentiated from hyperventilation syndrome by the presence of low SPO2 levels and lack of carpopedal spasms.
When handing off respiratory patients in the ED, saying “No shark fins were present,” or “CO2 waveforms were normal,” may be meaningless to staff unfamiliar with waveform capnography. Instead, put the information in context; provide a pre- and post-treatment PetCO2 reading just as you would a room air and post-treatment oxygen saturation, and say, “CO2 waveforms showed consistent alveolar emptying and no evidence of bronchospasm.”
Patients at risk of impaired perfusion
PetCO2 is an excellent non-invasive surrogate for measuring cardiac output. Since decreased PetCO2 levels often appear before frank hypotension, waveform capnography should be used on all patients at risk of impaired perfusion. In patients with risk of impaired perfusion, waveform morphology is not as important as the PetCO2.
In severe sepsis patients, decreased PetCO2 strongly correlates to elevated serum lactate levels, a reliable measure of metabolic stress. PetCO2 of less than 25 mm Hg is associated with serum lactate greater than 4 mmol/L, and these patients are at far greater risk of septic shock.
When handing off trauma patients or other patients at risk of hypoperfusion, inform the ED staff of current PetCO2 levels, and any post-treatment changes.
Patients at risk of CNS depression
Most of the sedatives and analgesics we administer can reduce respiratory drive, as can organic causes, trauma, and illicit drugs. Monitor PetCO2 in any patient with altered mental status, and be alert for hypoventilation. Re-administer naloxone if appropriate, or if the patient is arousable to verbal stimulus. The simple expedient of engaging your patient in conversation throughout transport can keep the patient from becoming somnolent and maintain PetCO2 at physiologically appropriate levels. If you administered naloxone, or inserted an invasive airway device for airway maintenance in the obtunded patient, document pre- and post-treatment PetCO2 and inform the ED staff of those readings.
Use these capnography practices to guide your patient care, paint a more comprehensive patient picture in your documentation, and better inform receiving hospital staff of critical information. As we better communicate capnography findings to hospital staff, more of them will begin to embrace the technology and value its findings.
References
- Corbo J, Bijur P, Lahn M, Gallagher EJ. Concordance Between Capnography and Arterial Blood Gas Measurements of Carbon Dioxide in Acute Asthma. Annals of Emergency Medicine. Volume 46, Issue 4, October 2005, Pages 323–327.
- What happens to PetCO2 When There Is a Pneumothorax
- Capnograph Waveforms In the Mechanically Ventilated Patient
- Ahrens T, Schallom L, Bettorf K, Ellner S, Hurt G, O’Mara V, Ludwig J, George W, Marino T, Shannon W. End-tidal Carbon Dioxide Measurements as a Prognostic Indicator of Outcome in Cardiac Arrest. American Journal of Critical Care. 2001 Nov;10(6):391-8.
- Callaham M, Barton C. Prediction of outcome of cardiopulmonary resuscitation from end-tidal carbon dioxide concentration. Critical Care Medicine. 1990 Apr;18(4):358-62.
- Morisaki H, Takino Y, Kobayashi H, Ando Y, Ichikizaki K. End-tidal Carbon Dioxide Concentration During Cardiopulmonary Resuscitation in Patients with Pre-hospital Cardiac Arrest. Masui. 1991 Jul;40(7):1048-51.
- Berg RA, Henry C, Otto CW, Sanders AB, Kern KB, Hilwig RW, Ewy GA. Initial End-tidal CO2 Is Markedly Elevated During Cardiopulmonary Resuscitation After Asphyxial Cardiac Arrest. Pediatric Emergency Care. 1996 Aug;12(4):245-8.
- Steedman DJ, Robertson CE. Measurement of End-tidal Carbon Dioxide Concentration During Cardiopulmonary Resuscitation. Archives of Emergency Medicine. 1990 Sep;7(3):129-34.
- Falk JL, Rackow EC, Weil MH. End-tidal carbon dioxide concentration during cardiopulmonary resuscitation. New England Journal of Medicine. 1988 Mar 10;318(10):607-11.